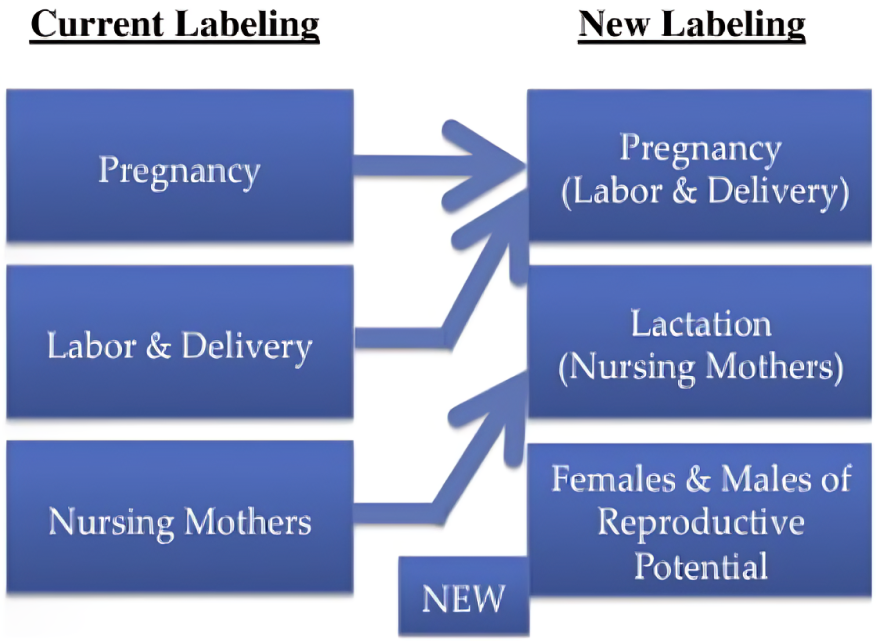
In 2015, the FDA replaced the outdated pregnancy risk categories (A, B, C, D, and X) on drug labels with a more comprehensive system.
The new labeling includes narrative sections for Pregnancy, Lactation, and Females and Males of Reproductive Potential.
This aims to provide detailed information on dosing, potential risks to the fetus, and other relevant considerations.
The change was prompted by the need for clearer information, as the old categories were deemed simplistic and often led to confusion.
The new system allows for better patient-specific counseling, though clinical interpretation remains necessary.
The FDA received input from various groups and conducted public hearings before implementing the changes.
Examples of drugs approved since 2015 with the new labeling include Addyi, Descovy, Entresto, Harvoni, and Praluent. The former categories (A, B, C, D, and X) are now considered outdated, with the new system providing more nuanced and detailed information about the risks and benefits of medication use during pregnancy.
To read more about the new system visit "https://www.drugs.com/pregnancy-categories.html" .